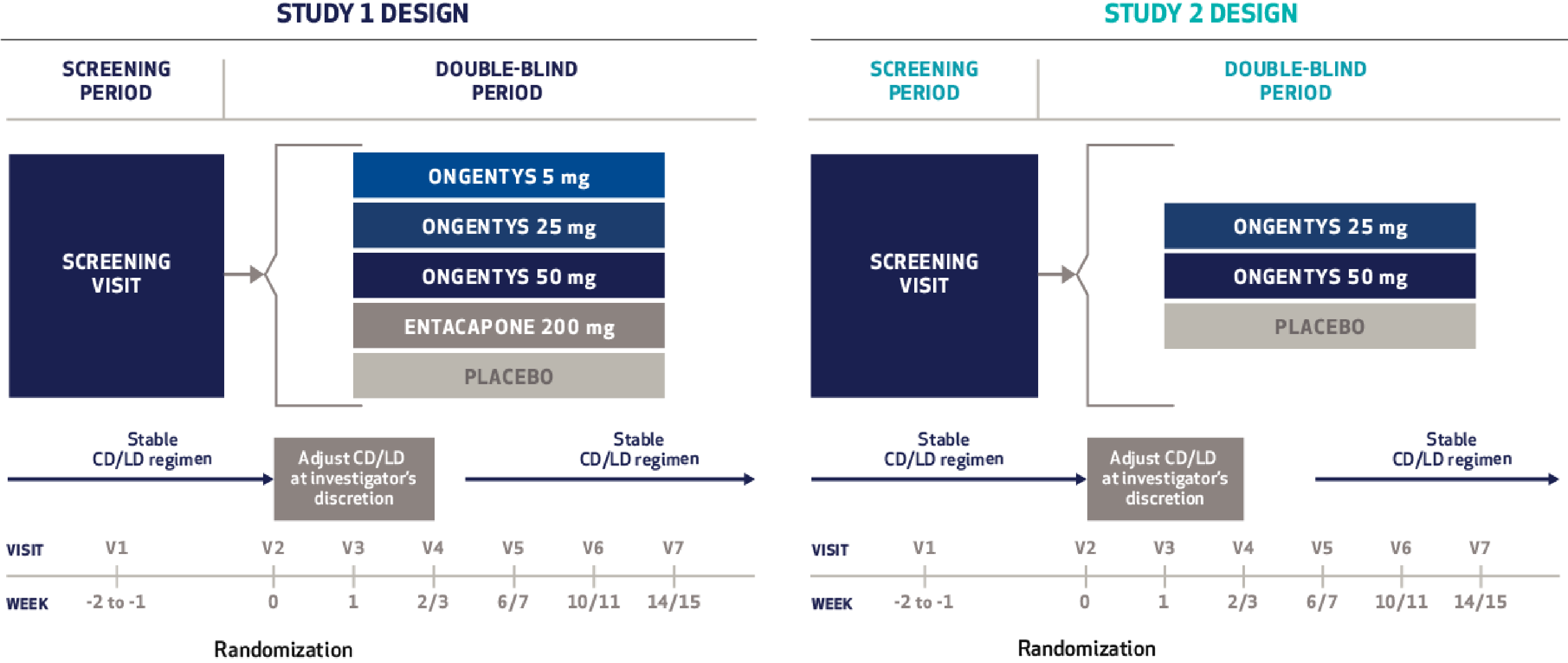
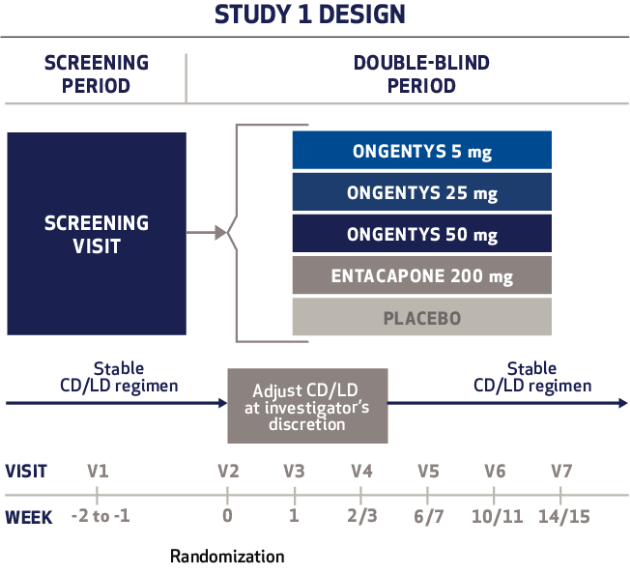
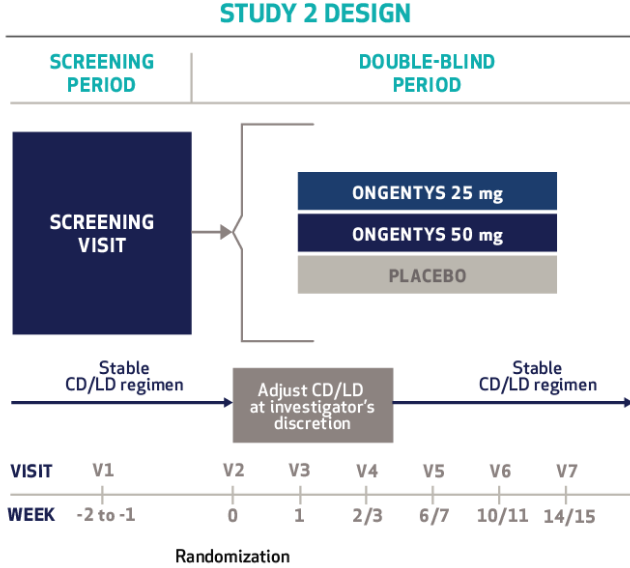
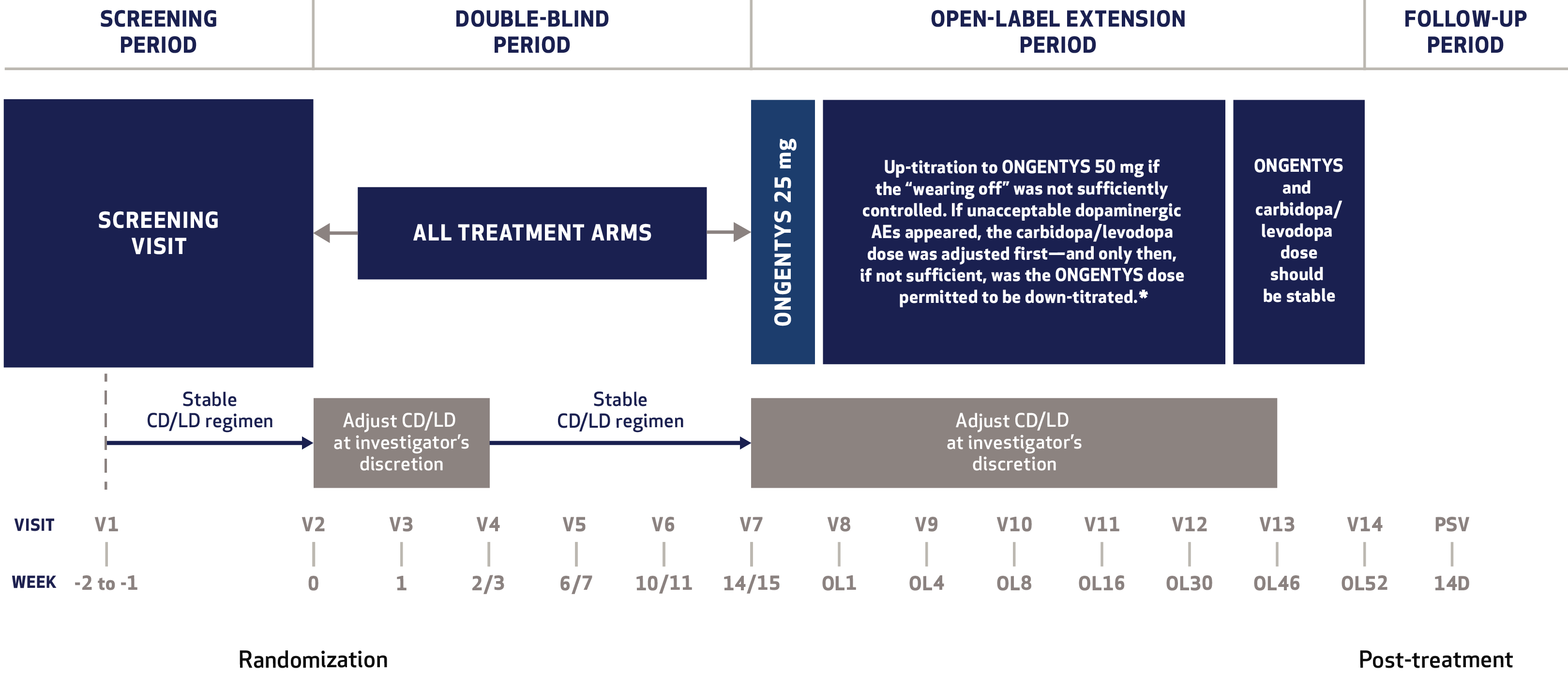
Cardiovascular Effects with Concomitant Use of Drugs Metabolized by Catechol-O-Methyltransferase (COMT) - Possible arrhythmias, increased heart rate, and excessive changes in blood pressure may occur with concomitant use of ONGENTYS and drugs metabolized by COMT, regardless of the route of administration (including inhalation). Monitor patients treated concomitantly with ONGENTYS and drugs metabolized by COMT.
Falling Asleep During Activities of Daily Living and Somnolence - Patients have reported falling asleep while engaged in activities of daily living, including driving, which may result in accidents. Consider discontinuing ONGENTYS or adjusting other dopaminergic/sedating medications. Advise patients to avoid driving and other potentially dangerous activities.
Hypotension/Syncope - Monitor patients for hypotension and advise patients about the risk for syncope. If necessary, consider discontinuing ONGENTYS or adjusting the dosage of other medications that can lower blood pressure.
Dyskinesia - ONGENTYS may cause or exacerbate dyskinesia. Consider levodopa or dopaminergic medication dose reduction.
Hallucinations and Psychosis - Consider stopping ONGENTYS if these occur. Patients with a major psychotic disorder should ordinarily not be treated with ONGENTYS.
Impulse Control/Compulsive Disorders - Patients may experience intense urges (eg, gambling, sexual, spending money, binge eating) and the inability to control them. It is important for prescribers to ask about the development of new or increased urges. Monitor for occurrence of intense urges and consider discontinuing ONGENTYS if they occur.
Withdrawal-Emergent Hyperpyrexia and Confusion - A symptom complex resembling neuroleptic malignant syndrome can develop with rapid dose reduction or withdrawal of drugs that increase central dopaminergic tone. When discontinuing ONGENTYS, monitor patients and consider adjustment of dopaminergic therapies as needed.
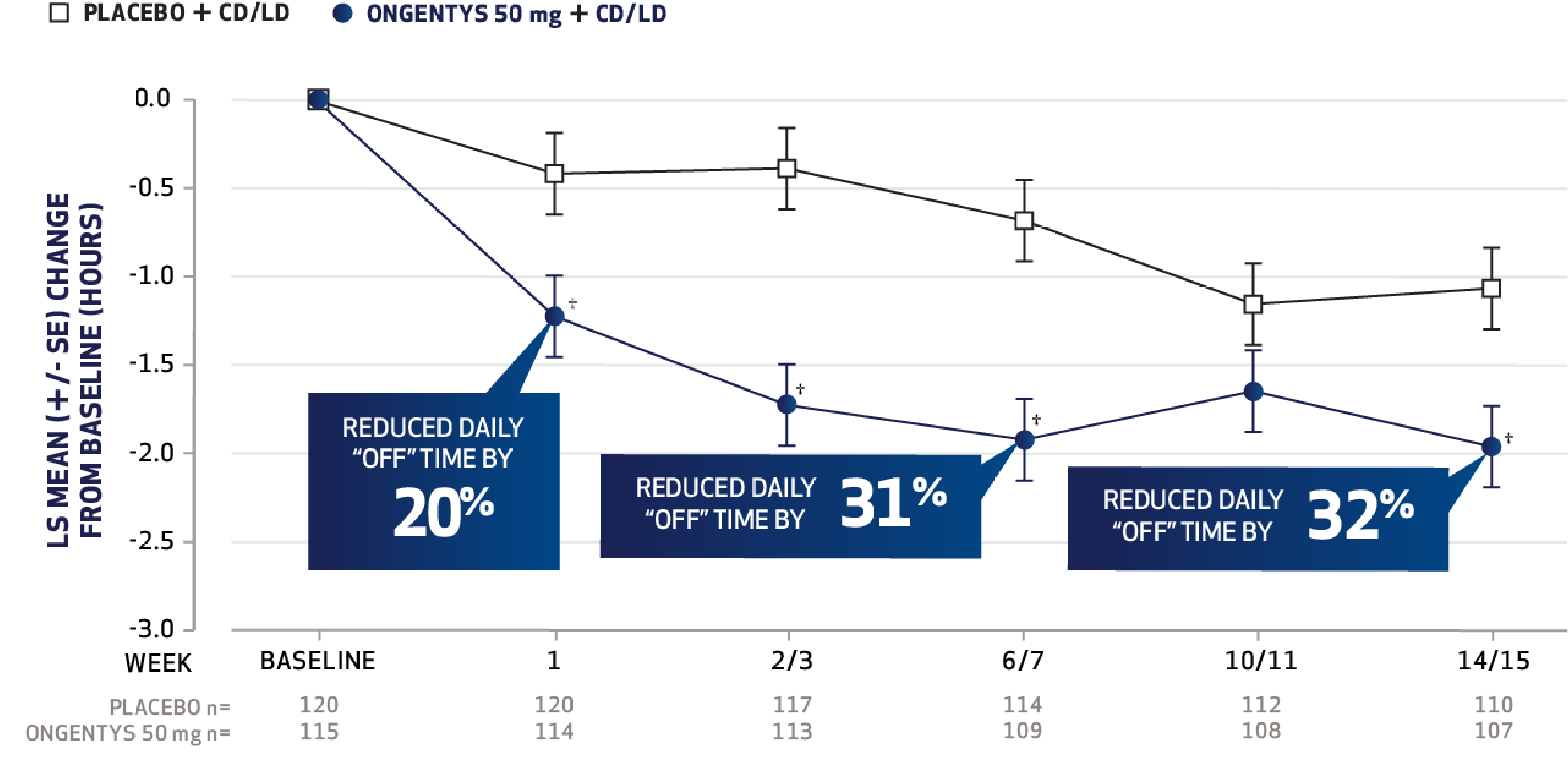
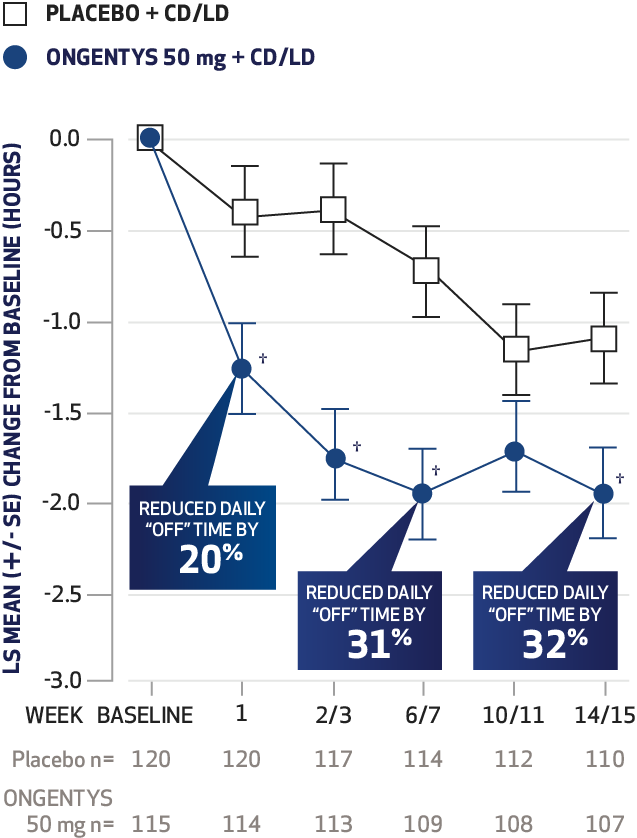
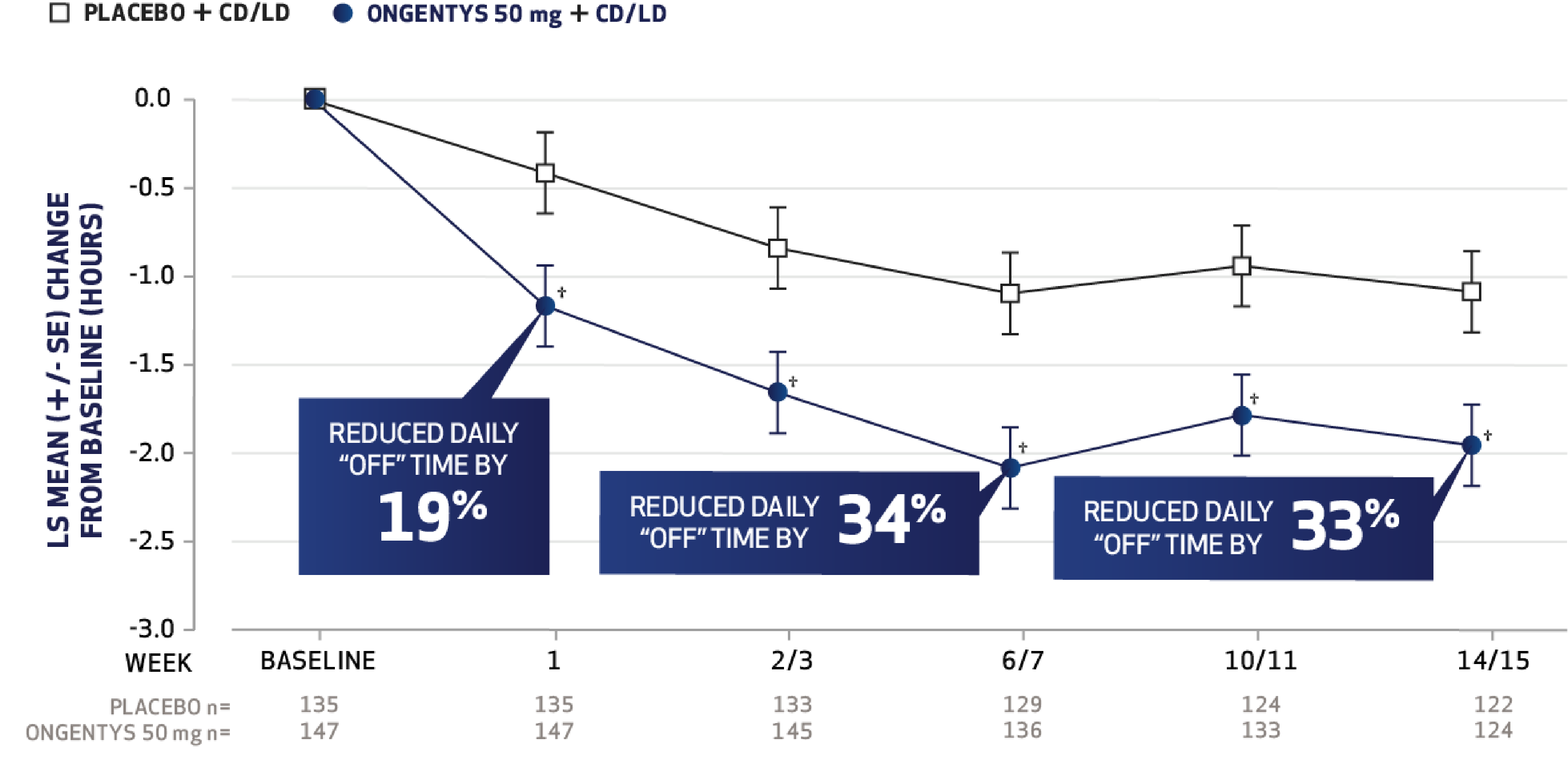
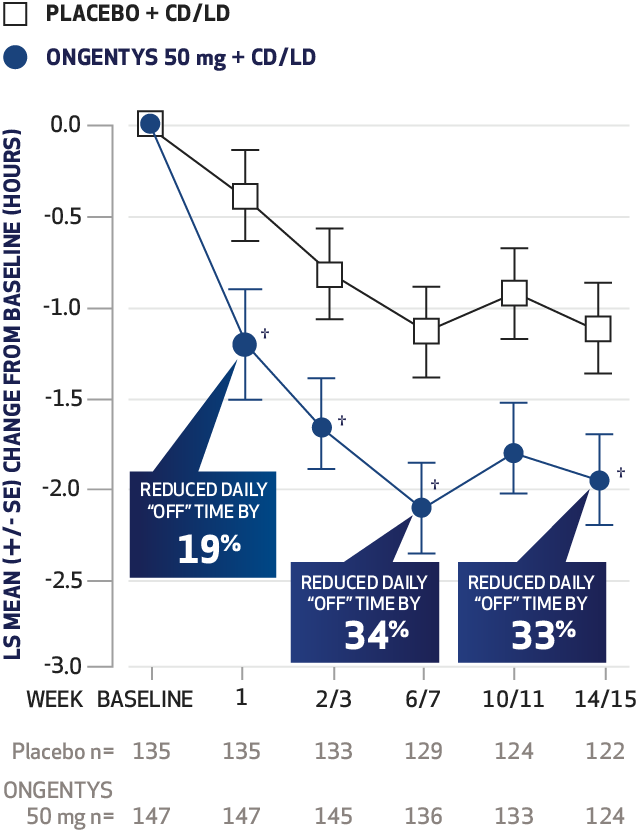
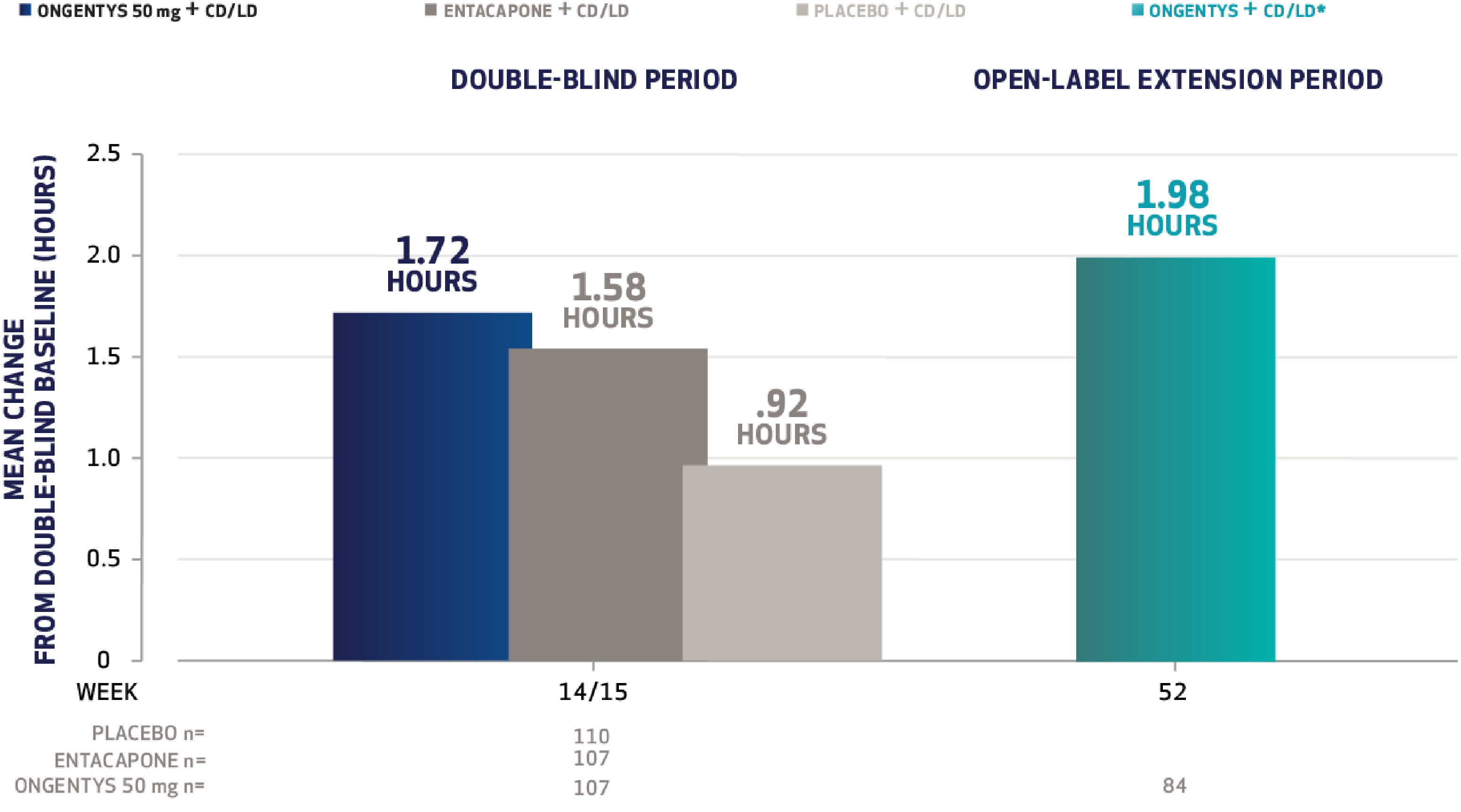
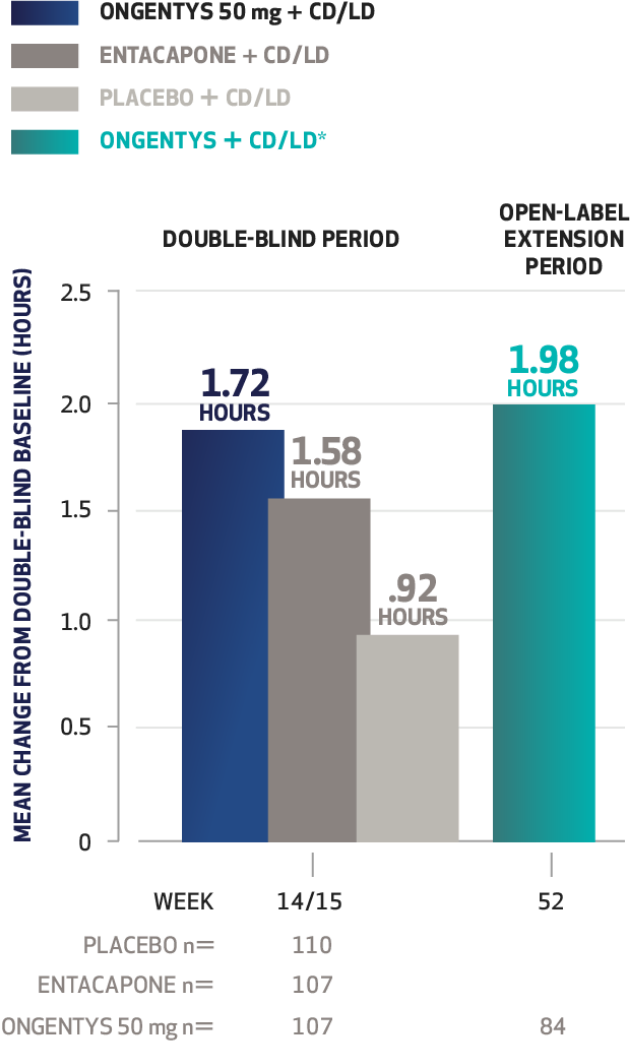
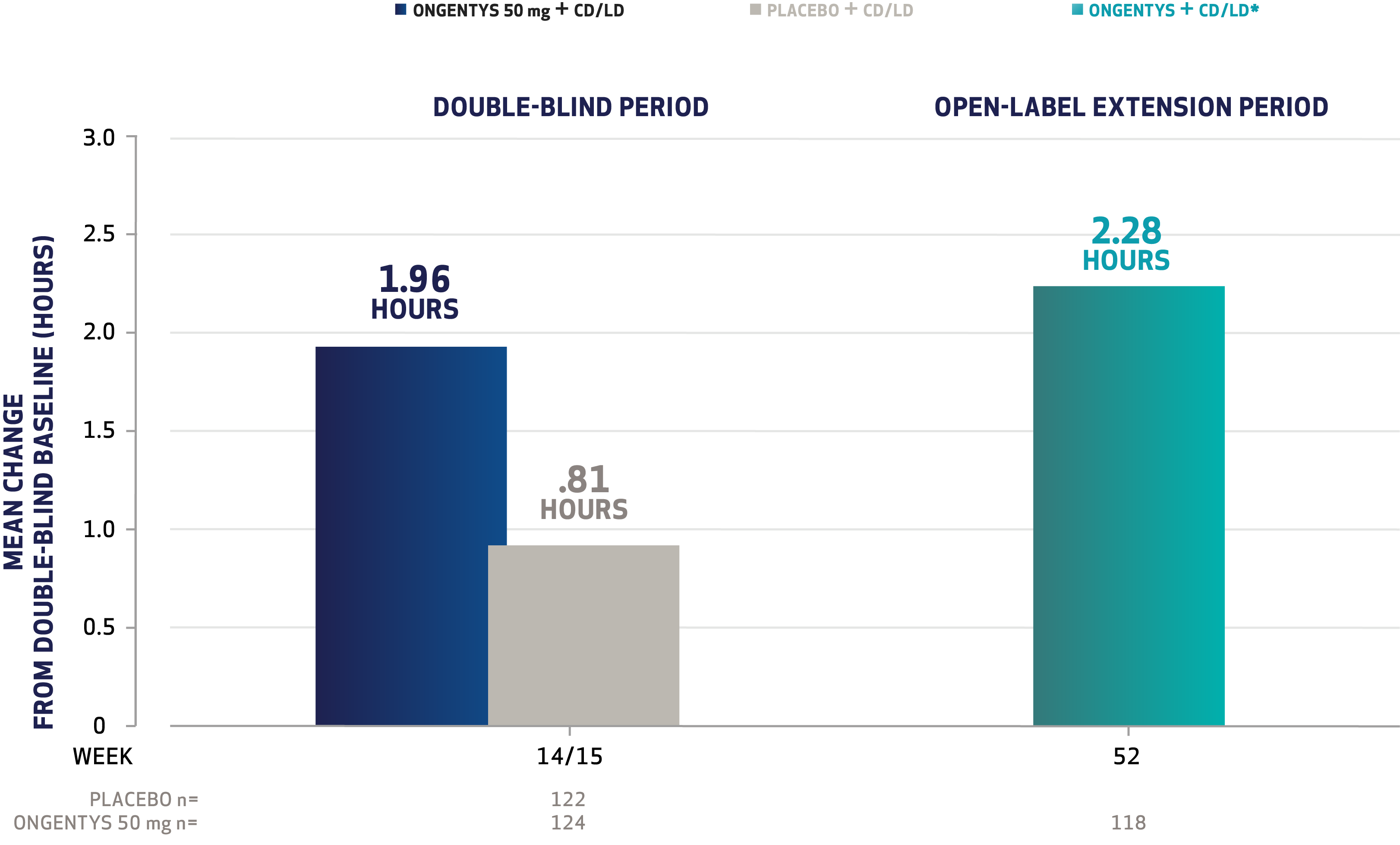
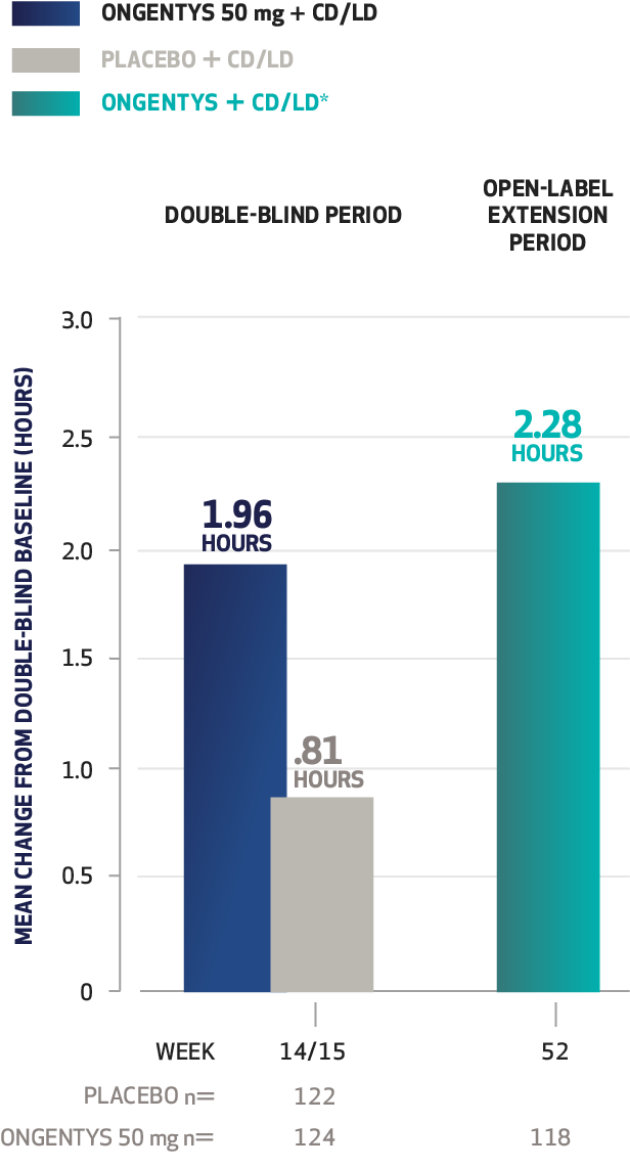
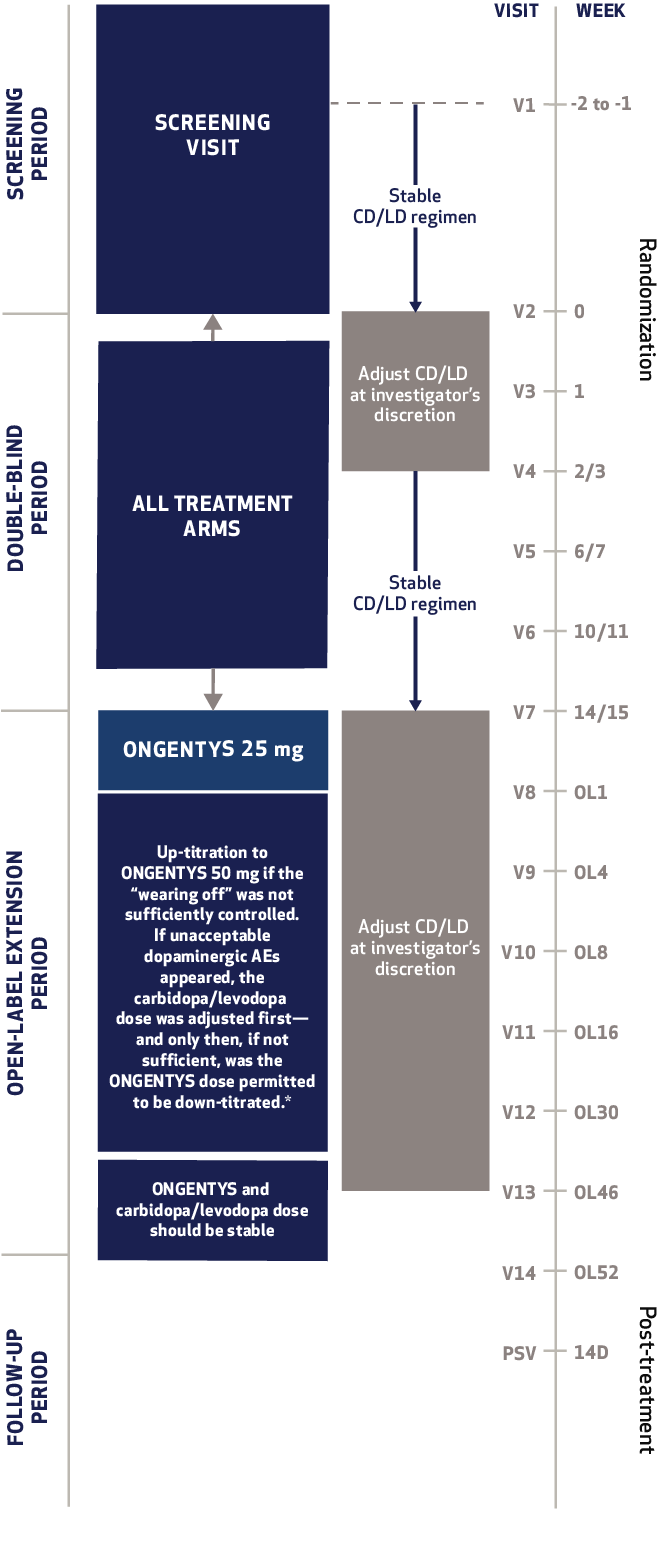
The most common adverse reactions (incidence at least 4% and greater than placebo) were dyskinesia, constipation, blood creatine kinase increased, hypotension/syncope, and weight decreased.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit MedWatch at or call .
Please see ONGENTYS full .
References:
- ONGENTYS [package insert]. Bridgewater, NJ: Amneal Pharmaceuticals LLC; 2021.
- Data on file. Amneal Pharmaceuticals LLC.
- Rocha JF, Eberbach G, Lees A, et al. The added benefit of opicapone when used early in Parkinson's disease patients with levodopa-induced motor fluctuations: a post-hoc analysis of BIPARK-I and -II. Front Neurol. 2021;12:754016.
- Reichmann H, Lees A, Rocha JF, Magalhães D, Soares-da-Silva P; Optipark Investigators. Effectiveness and safety of opicapone in Parkinson's disease patients with motor fluctuations: the OPTIPARK open-label study. Transl Neurodegener. 2020;9(1):9.
- Ferreira JJ, Lees A, Rocha JF, Poewe W, Rascol O, Soares-da-Silva P. Long-term efficacy of opicapone in fluctuating Parkinson's disease patients: a pooled analysis of data from two phase 3 clinical trials and their open-label extensions. Eur J Neurol. 2019:26(7):953-960.
- Ferreira JJ, Lees A, Rocha JF, et al. Opicapone as an adjunct to levodopa in patients with Parkinson's disease and end-of-dose motor fluctuations: a randomized, double-blind, controlled trial. Lancet Neurol. Published December 22, 2015. doi:10.1016/S1474-4422(15)00336-1
- Lees AJ, Ferreira J, Rascol O, et al; BIPARK-2 Study Investigators. Opicapone as adjunct to levodopa therapy in patients with Parkinson disease and motor fluctuations: a randomized clinical trial. JAMA Neurol. 2017;74(2):197-206.